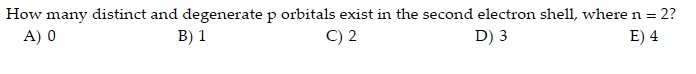

Here we propose a novel and realistic flat-band model based on threefold degenerate p-orbitals in two-dimensional ionic materials. Having said this, for most purposes the hydrogenic orbitals are very good approximations. Though several theoretical models have been proposed to design electronic flat-bands, the definite experimental realization in two-dimensional atomic crystal is still lacking. There is a single three electron wavefunction that can be approximately decomposed into hydrogenic 1s and 2s orbitals for convenience. For example in a lithium atom there is not actually two electrons in a 1s orbital and one in a 2s orbital. They should be regarded as a useful way of building up the complete electronic structure but they are not themselves real. The bonding orbitals >p2 and t 3 have the same energy and are said to be degenerate, as are the antibonding orbitals tf/4 and 5. (In O2 and F2 the order of : Figure 15.3 The six benzene tt molecular orbitals. In that case we do get hydrogenic type orbital as solutions, but we have to bear in mind that they are approximate solutions. The 2p, 2py, 2p atomic orbitals are degenerate in an atom and have been separated for convenience. The radial part of the orbitals will be different for different central potentials but the angular part is dictated by the spherical symmetry and is the same for all (smooth monotonic) central potentials.īut for any atom with more than two electrons the potential is not centrally symmetric because it includes terms like $1/r_$ terms tend to average out to a central force. Now, let us compare these p subshell degeneracies through a graphical representation. Electrons in 4p involve 4px, 4py, and 4pz, these also have a similar energy level as one another hence these are also degenerate. The 1s, 2s, etc orbitals are solutions for a central potential, and for any smooth monotonic central potential we'll get solutions of this form. These orbitals have similar energy levels, hence are called degenerate molecular orbitals. The remaining -orbitals transform as a triply degenerate representation.Polyelectronic atoms don't have atomic orbitals - though they are a very useful approximation for describing the properties of polyelectronic atoms. The and orbitals, which have lobes directed at ligands, become a higher-energy doublet which transforms as the representation of the octahedral group. However, in atoms with more than one electron, orbitals with different values of l for a given value for n are not degenerate. The octahedral crystal field partially breaks the five-fold symmetry of the -orbitals. In hydrogen, all orbitals with the same principal quantum number 'n' (1,2,3.) are degenerate, regardless of the orbital angular momentum quantum number 'l' (0,1.n-1 or s,p,d,). It usually refers to electron energy levels or sublevels. The effect of the ligands can be represented by a crystal field parameter, whose magnitude is typically in the range 7,000–30,000. Degenerate is used in quantum mechanics to mean of equal energy. A common configuration is the highly symmetrical octahedral complex, in which six equivalent ligands are bound to the central ion. Transition metals are likely to form compounds in which they are centrally bonded to several molecules or ions, known as ligands. Explanation: For example the px,py and pz electron orbitals of an atom exist in the directions of the x, y and z coordinates but are all of equivalent energy, so are said to be degenerate. numbers lead to the same energy state, the system is said to be degenerate. The positive and negative regions of the wavefunctions are shown in the blue and yellow spheres, respectively. Chemistry Quantum Mechanical Model of the Atom Orbitals, and Probability Patterns 1 Answer Simon Moore Orbitals with the same energy. These can be occupied by a maximum of 10 electrons. Transcribed image text: 02 Question (1 point) Consider the following atomic orbitals and the definition of the term 'degenerate.
Degenerate atomic orbitals free#
A free transition-metal atom or ion has five-fold degenerate -orbitals, pictured at the top of the graphic.
